Differentiated Thyroid Cancer
Initial Evaluation of Thyroid Nodules
Tables & Figures
Table 4. Familial & Heritable Forms of Thyroid Cancer
Familial Adenomatosis Polyposis
| Gene | APC |
| Benign Thyroid Disease | 40% |
| Cancer | 0.4–12% |
| Cancer Types | CMV-PTC: 63% FV-PTC: 25% PTC: 12% |
PTEN-Hamartoma Tumor (Cowden)
| Gene | PTEN |
| Benign Thyroid Disease | 75% |
| Cancer | 35% |
| Cancer Types | PTC: 50% FV-PTC: 28% FTC: 14% |
Carney Complex Type 1
| Gene | PRKARIA |
| Benign Thyroid Disease | ≤ 75% |
| Cancer | < 5% |
| Cancer Types | PTC FTC |
RET-Associated
| Gene | RET |
| Benign Thyroid Disease | — |
| Cancer | 100% |
| Cancer Types | MTC |
DICER1
| Gene | DICER1 |
| Benign Thyroid Disease | ≤ 30% |
| Cancer | — |
| Cancer Types | FTC FV-PTC |
CMV = Cribiform Morular Variant; FV = Follicular Variant
Percentages refer to disease prevalence.
Bethesda System
Bethesda Category I
| Meaning | Non Diagnostic |
| Estimated Risk of Malignancy | 5–10% |
Bethesda Category II
| Meaning | Benign |
| Estimated Risk of Malignancy | 0–3% |
Bethesda Category III
| Meaning | Atypia of Undetermined Significance (AUS) / Follicular Lesion of Undetermined Significance (FLUS) |
| Estimated Risk of Malignancy | 10–30% |
Bethesda Category IV
| Meaning | Suspicious for a Follicular (or Hürthle Cell) Neoplasm |
| Estimated Risk of Malignancy | 25–40% |
Bethesda Category V
| Meaning | Suspicious for Malignancy |
| Estimated Risk of Malignancy | 50–75% |
Bethesda Category VI
| Meaning | Malignant |
| Estimated Risk of Malignancy | 97–99% |
As reported in The Bethesda System by Cibas and Ali (DOI 10.1007/978-3-319-60570-8).
Initial Management
Tables & Figures
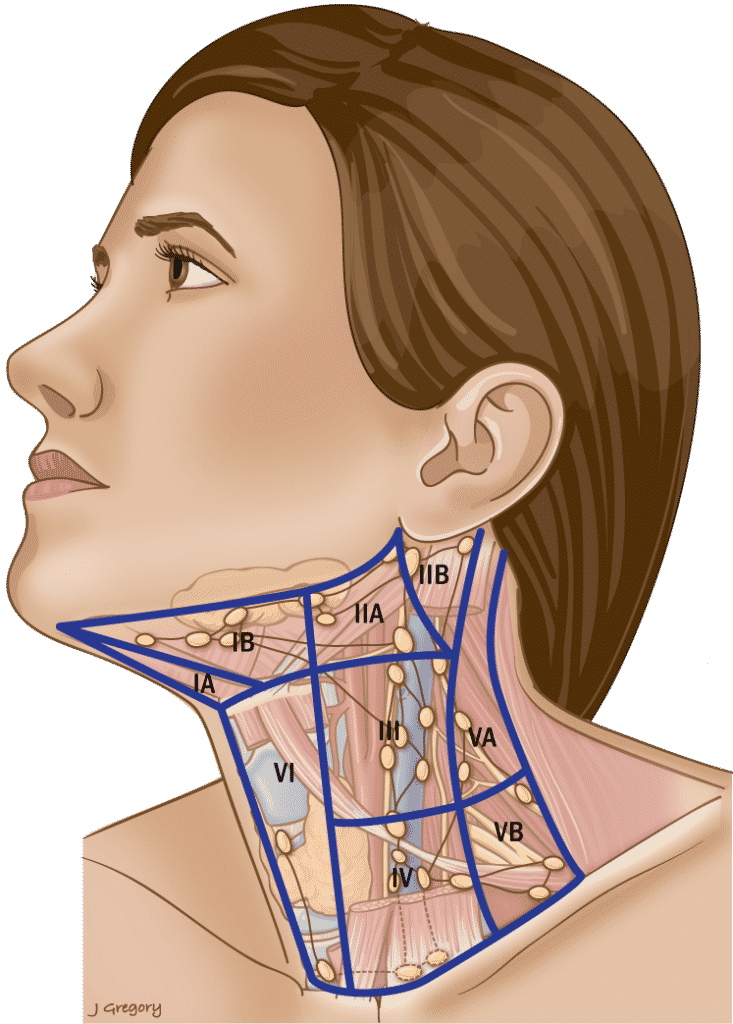
Table 3. Anatomic Boundaries of the Neck Levels
Level I
| Anterior |
| Anterior belly of the contralateral digastric muscle. |
| Posterior |
| Stylohyoid muscle |
| Superior |
| Body of the mandible |
| Inferior |
| Hyoid |
Triangular boundaries comprising anterior bellies of digastric muscles and hyoid separates IA & IB.
Level II
| Anterior |
| Stylohyoid muscle |
| Posterior |
| Posterior SCM |
| Superior |
| Skull base |
| Inferior |
| Hyoid |
CN XI separates IIA & IIB. IIA nodes lie anterior to IJV.
Level III
| Anterior |
| Sternohyoid muscle |
| Posterior |
| Stylohyoid muscle |
| Superior |
| Hyoid |
| Inferior |
| Horizontal plane defined by the cricoid cartilage. |
Level IV
| Anterior |
| Sternohyoid muscle |
| Posterior |
| Posterior SCM |
| Superior |
| Inferior border of the cricoid cartilage. |
| Inferior |
| Clavicle |
Level V
| Anterior |
| Posterior SCM |
| Posterior |
| Anterior border of trapezius. |
| Superior |
| Convergence of the SCM and trapezius. |
| Inferior |
| Clavicle |
Inferior border of cricoid separates VA & VB.
Level VI
| Anterior |
| Anterior layer of the cervical fascia. |
| Posterior |
| Deep layer of the cervical fascia. |
| Superior |
| Hyoid superiorly |
| Inferior |
| Sternal notch |
Level VII
| Anterior |
| Sternum |
| Posterior |
| Deep layer of the cervical fascia. |
| Superior |
| Sternal notch |
| Inferior |
| Innominate on right and equivalent plane on the left. |
SCM = sternocleidomastoid muscle
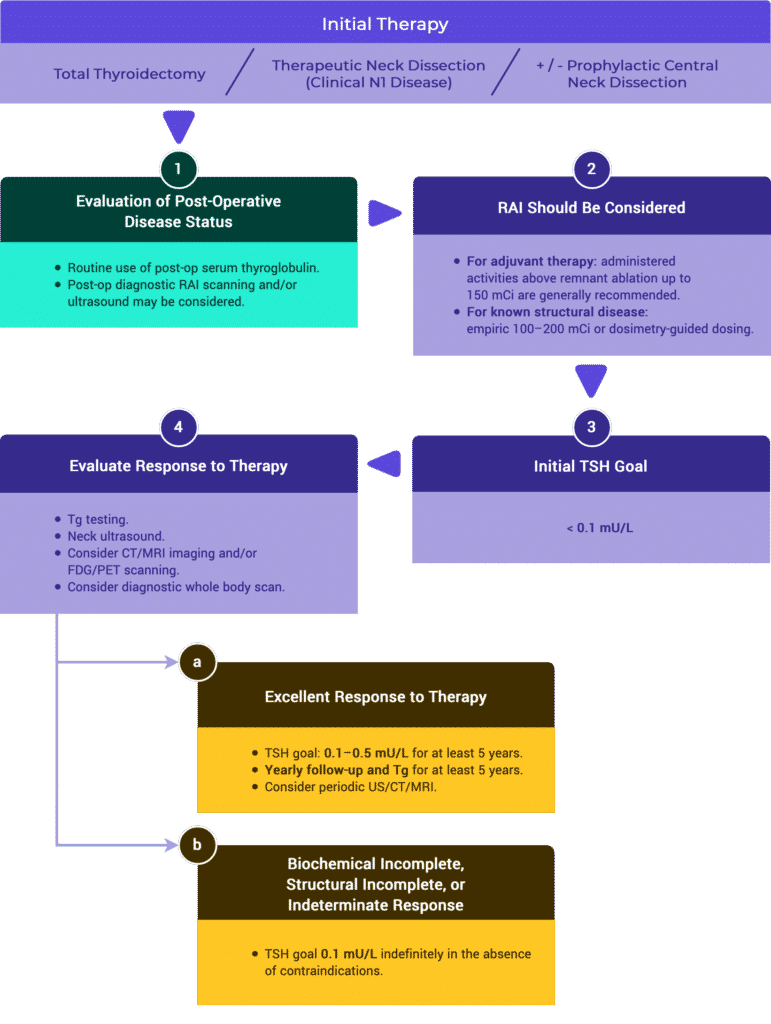
Initial Staging & Response to Therapy
Initial Staging
Tables & Figures
Response to Therapy
Management Recommendations Based on Risk Stratification
Tables & Figures
Table 11. Risk Stratification System with Proposed Modifications (ATA 2009)
Risk Level
ATA Low Risk
- Papillary thyroid cancer (with all of the following):
- No local or distant metastases,
- All macroscopic tumor has been resected,
- No tumor invasion of loco-regional tissues or structures,
- The tumor does not have aggressive histology (e.g. tall cell, hobnail variant, columnar cell carcinoma),
- If I131 is given, there are no RAI-avid metastatic foci outside the thyroid bed on the first post-treatment whole-body RAI scan,
- No vascular invasion,
- Clinical N0 or ≤ 5 pathologic N1 micrometastases (< 0.2 cm in largest dimension).*
- Intrathyroidal, encapsulated follicular thyroid variant of papillary thyroid cancer.*
- Intrathyroidal, well-differentiated follicular thyroid cancer with capsular invasion and no or minimal (< 4 foci) vascular invasion.*
- Intrathyroidal, papillary microcarcinoma, unifocal or multifocal, including BRAFV600E mutated (if known).*
ATA Intermediate Risk
- Microscopic invasion of tumor into the perithyroidal soft tissues.
- RAI-avid metastatic foci in the neck on the first post-treatment whole-body RAI scan.
- Aggressive histology (e.g. tall cell, hobnail variant, columnar cell carcinoma).
- Papillary thyroid cancer with vascular invasion.
- Clinical N1 or > 5 pathologic N1 with all involved lymph nodes < 3 cm in largest dimension.*
- Multifocal papillary microcarcinoma with ETE and BRAFV600E mutated (if known).*
ATA High Risk
- Macroscopic invasion of tumor into the perithyroidal soft tissues (gross ETE).
- Incomplete tumor resection.
- Distant metastases.
- Postoperative serum thyroglobulin suggestive of distant metastases.
- Pathologic N1 with any metastatic lymph node ≥ 3 cm in largest dimension.*
- Follicular thyroid cancer with extensive vascular invasion (> 4 foci of vascular invasion).*
Response to Therapy Based on Initial Management
Tables & Figures
Table 2a. Response to Therapy: Total Thyroidectomy & RRA (ATA compared to ESMO)
ATA // ESMO
Response to Therapy
Excellent
| Imaging |
| No relevant findings on imaging. |
| TSH-Suppressed Tg |
| < 0.2 ng/mL |
| TSH-Stim TG |
| < 1 ng/mL |
| TgAB |
| • Not specified (ATA) • Undetectable (ESMO) |
Biochem Incomplete
| Imaging |
| No relevant findings on imaging. |
| TSH-Suppressed Tg |
| 1 ng/mL |
| TSH-Stim TG |
| 10 ng/mL |
| TgAB |
| Rising TgAB levels |
Structural Incomplete
| Imaging |
| Evidence of structural or functional disease. * |
| TSH-Suppressed Tg |
| * |
| TSH-Stim TG |
| * |
| TgAB |
| * |
Indeterminate
| Imaging |
| Nonspecific findings on US/CT. — OR — Faint uptake in thyroid bed on RAI scanning. |
| TSH-Suppressed Tg |
| • Tg detectable, but < 1 ng/mL (ATA) • 0.2–1 ng/mL (ESMO) |
| TSH-Stim TG |
| • Stimulated Tg detectable, but < 10 ng/mL (ATA) • 1–10 ng/mL (ESMO) |
| TgAB |
| TgAB levels stable or declining in the absence of structural or functional disease. |
* Regardless of Tg or TgAB levels.
Table 2b. Response to Therapy: Total Thyroidectomy Alone
ESMO
Response to Therapy
Excellent
| Imaging |
| No relevant findings on imaging. |
| TSH-Suppressed Tg |
| < 0.2 ng/mL |
| TSH-Stim TG |
| Not Specified |
| TgAB |
| Undetectable |
Biochem Incomplete
| Imaging |
| No relevant findings on imaging. |
| TSH-Suppressed Tg |
| Tg > 5 ng/mL – OR – Rising Tg values with similar TSH levels. |
| TSH-Stim TG |
| — |
| TgAB |
| Rising TgAB levels |
Structural Incomplete
| Imaging |
| Evidence of structural or functional disease. * |
| TSH-Suppressed Tg |
| * |
| TSH-Stim TG |
| * |
| TgAB |
| * |
Indeterminate
| Imaging |
| Nonspecific findings on US/CT. — OR — Faint uptake in thyroid bed on RAI scanning. |
| TSH-Suppressed Tg |
| < 0.2–5 ng/mL |
| TSH-Stim TG |
| Not Specified |
| TgAB |
| TgAB levels stable or declining in the absence of structural or functional disease. |
* Regardless of Tg or TgAB levels.
Table 2c. Response to Therapy: Lobectomy Alone
ESMO
Response to Therapy
Excellent
| Imaging |
| No relevant findings on imaging. |
| TSH-Suppressed Tg |
| Stable |
| TSH-Stim TG |
| Stable |
| TgAB |
| Undetectable |
Biochem Incomplete
| Imaging |
| No relevant findings on imaging. |
| TSH-Suppressed Tg |
| Rising Tg values with similar TSH levels |
| TSH-Stim TG |
| TgAB |
| Rising |
Structural Incomplete
| Imaging |
| Evidence of structural or functional disease. * |
| TSH-Suppressed Tg |
| * |
| TSH-Stim TG |
| * |
| TgAB |
| * |
Indeterminate
| Imaging |
| Nonspecific findings on US/CT. |
| TSH-Suppressed Tg |
| * |
| TSH-Stim TG |
| * |
| TgAB |
| * |
* Regardless of Tg or TgAB levels.
Genetic Alterations & Histologic Features
Tables & Figures
Table 7. Gene Alterations & Histology
Phenotype
Toxic Adenoma
Known Somatic Genetic Mutation / Alterations
- TSH-R
- GNAS
Benign Thyroid Nodules
Known Somatic Genetic Mutation / Alterations
- N-, H- and K-RAS
- EIF1AX
Noninvasive Follicular Tumor with Papillary-like Features (NIFTP)
Known Somatic Genetic Mutation / Alterations
- N-, H- and K-RAS
- BRAF K601E
Infiltrative Follicular Variant Papillary Thyroid Carcinoma (FVPTC)
Known Somatic Genetic Mutation / Alterations
- N-, H- and K-RAS
- BRAF V600E
Papillary Thyroid Carcinoma (PTC)
Known Somatic Genetic Mutation / Alterations
- RET/PTC
- BRAF V600E
- N-, H-, K-RAS
- TERT
Columnar Cell, Tall Cell, Hobnail Variant PTC
Known Somatic Genetic Mutation / Alterations
- BRAF V600E
Diffuse-sclerosing Variant PTC
Known Somatic Genetic Mutation / Alterations
- RET/PTC
Follicular Thyroid Carcinoma (FTC)
Known Somatic Genetic Mutation / Alterations
- N-, H- and K-RAS
- PAX8/PPAR
- PTEN
Hürthle Cell Carcinoma
Known Somatic Genetic Mutation / Alterations
- NRAS
- Genes in the PI3K-Akt Pathway
Poorly Differentiated Thyroid Carcinoma (PDTC)
Known Somatic Genetic Mutation / Alterations
- N-RAS (Insular)
- BRAF V600E
- PIK3CA
- RET/PTC
- TERT
Anaplastic Thyroid Carcinoma (ATC)
Known Somatic Genetic Mutation / Alterations
- N-RAS, BRAF V600E
- PIK3CA
- TP53
- β-catenin
- EIF1AX
Medullary Thyroid Carcinoma (MTC)
Known Somatic Genetic Mutation / Alterations
- RET (Germ Line Mutation in Inherited MTC, Somatic Mutation)
- N-, H- and K-RAS (Somatic Mutations)
Adapted with permission from Wolters Kluwer Health, Inc.: the author(s), titles of article, title of journal, volume number, issue number, inclusive pages and website URL to the journal page.
Table 8. Histologic Subtypes of PTC
Subtype
Follicular-variant
| Nuclear Features of PTC Present? |
| Yes |
| Characteristics |
| • RAS mutation more common. • Follicular growth pattern. |
| Prognosis |
| 10-year DSS 93% |
Follicular-variant Encapsulated, Invasive
| Nuclear Features of PTC Present? |
| — |
| Characteristics |
| • RAS mutation or PPARG rearrangement more common. • Low rate of LNM. |
| Prognosis |
| 10-year DSS ~100% |
Follicular-variant Nonencapsulated / Infiltrative
| Nuclear Features of PTC Present? |
| — |
| Characteristics |
| BRAF V600E more common. |
| Prognosis |
| Equivalent to classic / conventional PTC |
Columnar Cell
| Nuclear Features of PTC Present? |
| No |
| Characteristics |
| • Papillae lined by columnar cells with nuclear stratification. • Large tumors with capsular invasion are associated with LNM and DM. |
| Prognosis |
| Variable |
Cribiform Morular
| Nuclear Features of PTC Present? |
| Occasionally |
| Characteristics |
| • Presence of morules—squamoid areas with intranuclear inclusions and nuclear clearing. • Associated with Familial Adenomatosis Polyposis Syndrome. |
| Prognosis |
| Equivalent to classic / conventional PTC |
Classic / Conventional
| Nuclear Features of PTC Present? |
| Yes |
| Characteristics |
| LNM Common |
| Prognosis |
| • 5-year DSS 97.4% • 10-year DSS 93% |
Diffuse Sclerosing
| Nuclear Features of PTC Present? |
| Yes |
| Characteristics |
| • Diffuse fibrosis. • Dense lymphoid infiltration. • Squamous metaplasia. |
| Prognosis |
| • 5-year DSS 96% • Equivalent to high-risk PTC |
Tall Cell
| Nuclear Features of PTC Present? |
| Yes |
| Characteristics |
| • Extrathyroidal extension and LNM possible. • > 30% cells are 2x tall as wide and eosinophilic cytoplasm. |
| Prognosis |
| 5-year DSS 95.6% |
Hobnail
| Nuclear Features of PTC Present? |
| Yes |
| Characteristics |
| • Increased risk of DM. • > 30% have hobnail features (eccentric nuclei and tapering cytoplasm). • Syncytial or micropapillary clusters with apically placed nuclei. • BRAF V600E or p53 positive |
| Prognosis |
| 5-year DSS 83% |
Adapted with permission from Wolters Kluwer Health, Inc.: the author(s), titles of article, title of journal, volume number, issue number, inclusive pages and website URL to the journal page.
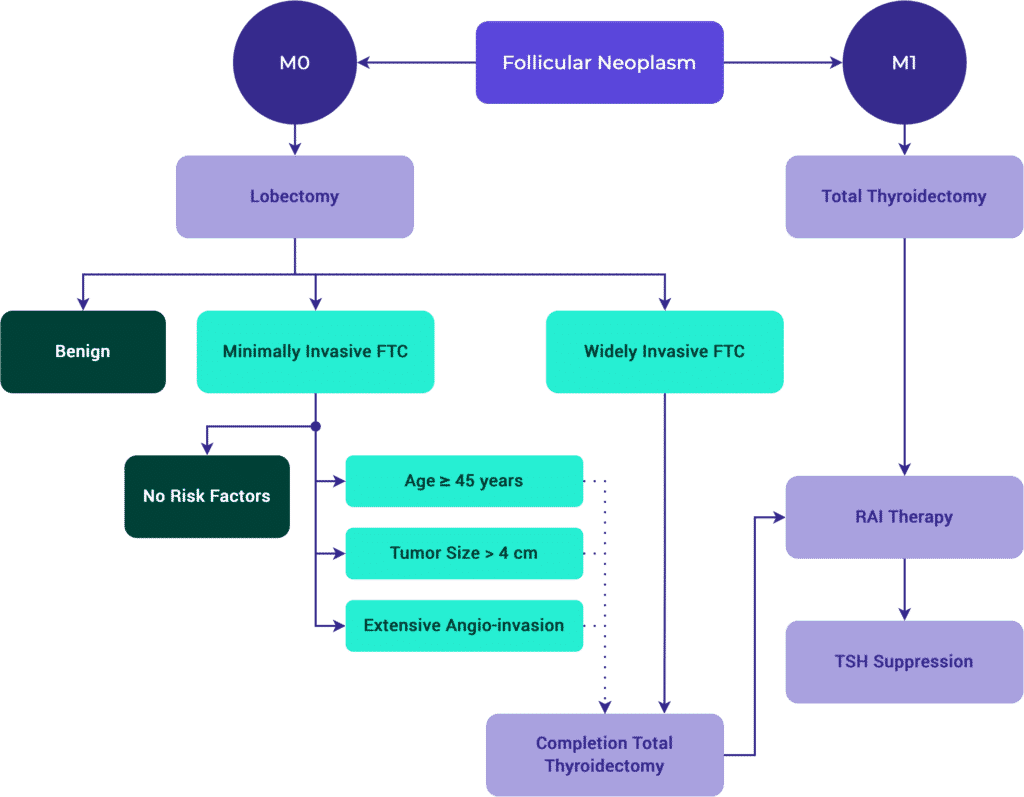
Use of Radioactive Iodine
Tables & Figures
Table 9. Remnant Ablation Decision Making
Tumor Description
Tumor ≤ 1 cm
| ATA Risk | Low |
| Disease specific survival improved by RAI | No |
| Disease free survival improved by RAI | No* |
| RAI indicated | No |
Tumor > 1–4 cm
| ATA Risk | Low |
| Disease specific survival improved by RAI | No |
| Disease free survival improved by RAI | Unclear data* |
| RAI indicated | Not routinely |
Tumor > 4 cm
| ATA Risk | Low |
| Disease specific survival improved by RAI | Unclear data* |
| Disease free survival improved by RAI | Unclear data* |
| RAI indicated | Consider |
Micro ETE (Any tumor size)
| ATA Risk | Low / Intermediate |
| Disease specific survival improved by RAI | No |
| Disease free survival improved by RAI | Unclear data* |
| RAI indicated | Consider |
Central LN Mets
| ATA Risk | Low / Intermediate |
| Disease specific survival improved by RAI | No |
| Disease free survival improved by RAI | Unclear data* |
| RAI indicated | Consider |
Lateral or Mediastinal LN Mets
| ATA Risk | Low / Intermediate |
| Disease specific survival improved by RAI | No |
| Disease free survival improved by RAI | Unclear data* |
| RAI indicated | Consider |
Any Size Gross ETE
| ATA Risk | High |
| Disease specific survival improved by RAI | Yes |
| Disease free survival improved by RAI | Yes* |
| RAI indicated | Yes |
Distant Metastasis
| ATA Risk | High |
| Disease specific survival improved by RAI | Yes |
| Disease free survival improved by RAI | Yes* |
| RAI indicated | Yes |
* Conflicting observational data.
The publisher for this copyrighted material is Mary Ann Liebert, Inc. publishers. This table is reprinted and adapted from THYROID.
Medullary Thyroid Cancer
Initial Evaluation of Thyroid Nodules Suspicious of MTC
Tables & Figures
Table 12. Most Common RET Mutations with Associated Risks
RET Mutation
Highest Risk Level (HST)
M918T
| RET Exon | 16 |
| Risk of Aggressive MTC | Highest |
| Incidence of Pheochromocytoma | 50% |
| Incidence of Hyperparathyroidism | – |
High Risk Level (H)
C634F/G/R/S/W/Y
| RET Exon | 11 |
| Risk of Aggressive MTC | High |
| Incidence of Pheochromocytoma | 50% |
| Incidence of Hyperparathyroidism | 20–30% |
A883F
| RET Exon | 15 |
| Risk of Aggressive MTC | High |
| Incidence of Pheochromocytoma | 50% |
| Incidence of Hyperparathyroidism | – |
Moderate Risk Level (MOD)
G533C
| RET Exon | 8 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10% |
| Incidence of Hyperparathyroidism | – |
C609F/G/R/S/Y
| RET Exon | 10 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10–30% |
| Incidence of Hyperparathyroidism | 10% |
C611F/G/S/Y/W
| RET Exon | 10 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10–30% |
| Incidence of Hyperparathyroidism | 10% |
C618F/R/S
| RET Exon | 10 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10–30% |
| Incidence of Hyperparathyroidism | 10% |
C620F/R/S
| RET Exon | 10 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10–30% |
| Incidence of Hyperparathyroidism | 10% |
C630R/Y
| RET Exon | 11 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10–30% |
| Incidence of Hyperparathyroidism | 10% |
D631Y
| RET Exon | 11 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 50% |
| Incidence of Hyperparathyroidism | – |
K666E
| RET Exon | 11 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10% |
| Incidence of Hyperparathyroidism | – |
E768D
| RET Exon | 13 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | – |
| Incidence of Hyperparathyroidism | – |
L790F
| RET Exon | 13 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10% |
| Incidence of Hyperparathyroidism | – |
V804L/M
| RET Exon | 14 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10% |
| Incidence of Hyperparathyroidism | 10% |
S891A
| RET Exon | 15 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10% |
| Incidence of Hyperparathyroidism | 10% |
R912P
| RET Exon | 16 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | – |
| Incidence of Hyperparathyroidism | – |
Table adapted from Table 4, Wells et al, Revised ATA guidelines for the management of medullary thyroid carcinoma, Thyroid 2015. The publisher for this copyrighted material is Mary Ann Liebert, Inc. publishers.
Management of Inherited Forms
Tables & Figures
Figure 12. ATA Management of Patients with RET Germline Mutation Detected on Genetic Screening.
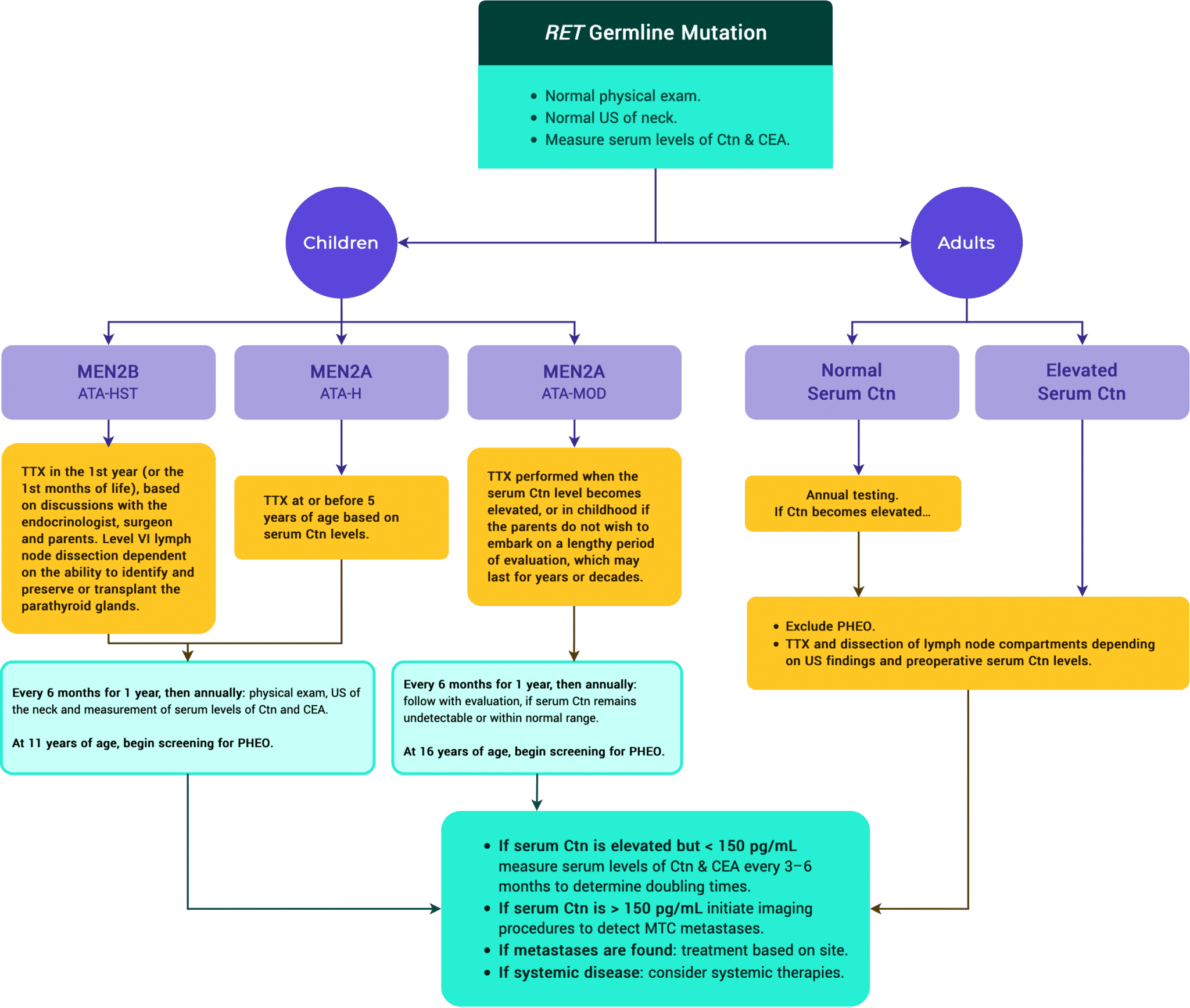
Table 12. Most Common RET Mutations with Associated Risks
RET Mutation
Highest Risk Level (HST)
M918T
| RET Exon | 16 |
| Risk of Aggressive MTC | Highest |
| Incidence of Pheochromocytoma | 50% |
| Incidence of Hyperparathyroidism | – |
High Risk Level (H)
C634F/G/R/S/W/Y
| RET Exon | 11 |
| Risk of Aggressive MTC | High |
| Incidence of Pheochromocytoma | 50% |
| Incidence of Hyperparathyroidism | 20–30% |
A883F
| RET Exon | 15 |
| Risk of Aggressive MTC | High |
| Incidence of Pheochromocytoma | 50% |
| Incidence of Hyperparathyroidism | – |
Moderate Risk Level (MOD)
G533C
| RET Exon | 8 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10% |
| Incidence of Hyperparathyroidism | – |
C609F/G/R/S/Y
| RET Exon | 10 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10–30% |
| Incidence of Hyperparathyroidism | 10% |
C611F/G/S/Y/W
| RET Exon | 10 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10–30% |
| Incidence of Hyperparathyroidism | 10% |
C618F/R/S
| RET Exon | 10 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10–30% |
| Incidence of Hyperparathyroidism | 10% |
C620F/R/S
| RET Exon | 10 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10–30% |
| Incidence of Hyperparathyroidism | 10% |
C630R/Y
| RET Exon | 11 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10–30% |
| Incidence of Hyperparathyroidism | 10% |
D631Y
| RET Exon | 11 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 50% |
| Incidence of Hyperparathyroidism | – |
K666E
| RET Exon | 11 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10% |
| Incidence of Hyperparathyroidism | – |
E768D
| RET Exon | 13 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | – |
| Incidence of Hyperparathyroidism | – |
L790F
| RET Exon | 13 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10% |
| Incidence of Hyperparathyroidism | – |
V804L/M
| RET Exon | 14 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10% |
| Incidence of Hyperparathyroidism | 10% |
S891A
| RET Exon | 15 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | 10% |
| Incidence of Hyperparathyroidism | 10% |
R912P
| RET Exon | 16 |
| Risk of Aggressive MTC | Moderate |
| Incidence of Pheochromocytoma | – |
| Incidence of Hyperparathyroidism | – |
Table adapted from Table 4, Wells et al, Revised ATA guidelines for the management of medullary thyroid carcinoma, Thyroid 2015. The publisher for this copyrighted material is Mary Ann Liebert, Inc. publishers.
Anaplastic Thyroid Cancer
Tables & Figures
Table 16. Panel of Routine Immunohistochemical Markers
IHC Marker
Pan-cytokeratins
| DTC | +++ |
| PDTC | +++ |
| ATC | +++/– |
| MTC | +++ |
| SCC | +++ |
| Lymphoma | – |
Thyroglobulin
| DTC | +++ |
| PDTC | +/– |
| ATC | – |
| MTC | – |
| SCC | – |
| Lymphoma | – |
Thyroid-transcription factor 1
| DTC | +++ |
| PDTC | +/– |
| ATC | –/+ |
| MTC | +/– |
| SCC | – |
| Lymphoma | – |
BRAFV600E
| DTC | +/– |
| PDTC | –/+ |
| ATC | –/+ |
| MTC | – |
| SCC | – |
| Lymphoma | – |
PAX8
| DTC | +++ |
| PDTC | +++ |
| ATC | +/– |
| MTC | +/– |
| SCC | – |
| Lymphoma | +/–a |
Ki-67b
| DTC | < 5% |
| PDTC | 5–30% |
| ATC | > 30% |
| MTC | < 20% |
| SCC | > 30% |
| Lymphoma | variable |
Chromogranin
| DTC | – |
| PDTC | – |
| ATC | – |
| MTC | +++ |
| SCC | – |
| Lymphoma | – |
Calcitonin
| DTC | – |
| PDTC | – |
| ATC | – |
| MTC | +++/– |
| SCC | – |
| Lymphoma | – |
Carcinembryonic antigen
| DTC | – |
| PDTC | – |
| ATC | – |
| MTC | +++ |
| SCC | – |
| Lymphoma | – |
p53
| DTC | – (rare +) |
| PDTC | –/+ |
| ATC | +/– |
| MTC | – |
| SCC | +/– |
| Lymphoma | +/– |
CD45, other lymphoid markers
| DTC | – |
| PDTC | – |
| ATC | – |
| MTC | – |
| SCC | – |
| Lymphoma | +++ |
“+” = relative positive staining; “–” = negative staining; “+/–” = variable positivity.
a PAX8 antibodies can cross-react with PAX5, which is expressed in lymphoid cells.
b Percentage of nuclei positive for Ki-67.
DTC = differentiated thyroid cancer; IHC = immunohistochemistry; MTC = medullary thyroid cancer; PDTC = poorly-differentiated thyroid cancer.
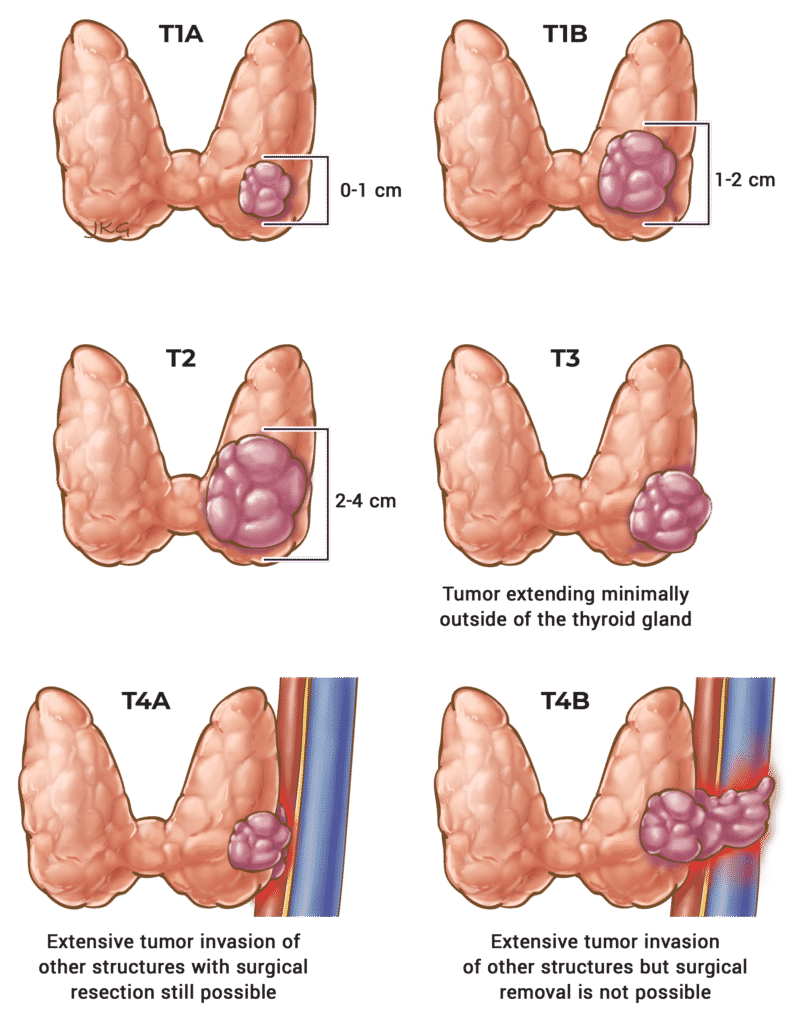
Table 13. T Staging for ATC
T Stage
T1
Description
< 2 cm
T2
Description
> 2 cm and < 4 cm
T3a
Description
> 4 cm
T3b
Description
Invasion of the strap muscles.
T4a
Description
Any size cancer with invasion of the larynx, trachea, esophagus, or the recurrent laryngeal nerve.
T4b
Description
Any size cancer with extension to the vertebrae or involvement of the carotid artery.
Adapted from the 8th edition AJCC.
Table 14. Prognostic Staging for ATC
Figure 13. Treatment Algorithm of ATC
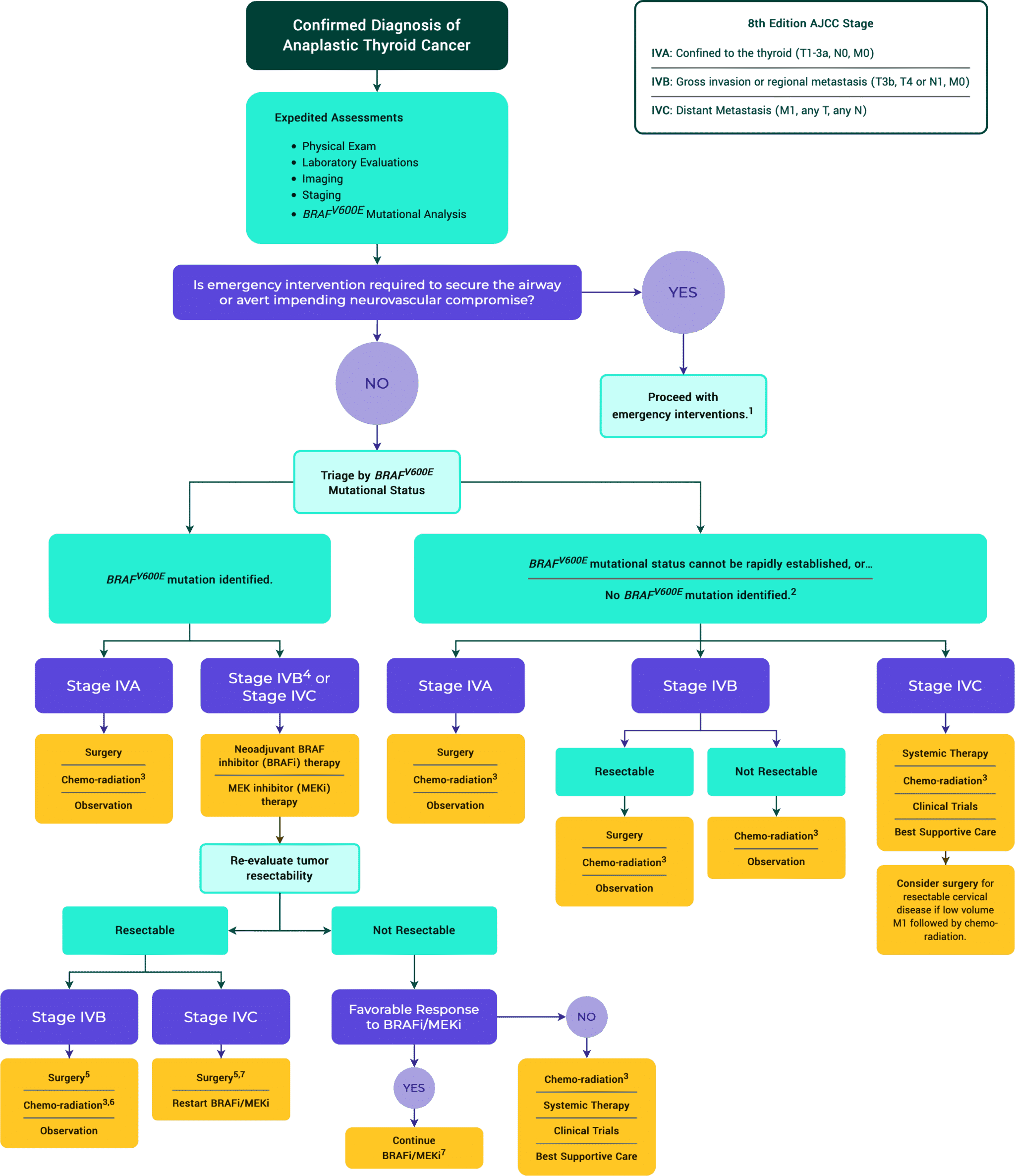
2 Next generation sequencing to identify targetable mutations is recommended. Specific inhibitors of oncogenic ALK, NTRK or RET fusion mutations can be considered, preferably within the context of a clinical trial.
3 Chemo-radiation: Typically, intensity-modulated radiation therapy is given to 70 Gy to gross tumor, 60–66 Gy to the post-op-bed and 50–54 Gy to potential microscopic residual disease regions with standard daily fractionation. The dose of radiation can change if clinically indicated. QUAD shot RT regimen can also be considered, typically for M1 disease, (3.7Gy BID x 2 days, can give up to 4–6 cycles, allow 1–4 weeks in between cycle pending on clinical scenario). At MSKCC, doxorubicin 20 mg/m2 weekly or paclitaxel 50 mg/m2 weekly is used throughout the radiation course but there are other reasonable regimens described in the literature.
4 If resectable, surgery followed by chemo-radiation can be considered as an alternative to neoadjuvant BRAFi/MEKi.
5 Discontinue BRAF inhibitor/MEK inhibitor therapy 2 days before surgery and resume 2–3 days after surgery.
6 Discontinue BRAF inhibitor/MEK inhibitor therapy 2 days before initiation of chemo-radiation.
7 Consider radiation therapy if it can be done without causing a significant delay in BRAF inhibitor/MEK inhibitor therapy.
Table 17. Mutations & Clinical Implications.
Mutations
BRAFV600E
Clinical Implications
Commonly seen in DTC which may be precursor to ATC.
Incidence in ATC
40–70% of ATCs and often seen in ATC arising from PTC.
RAS
Clinical Implications
Commonly seen in DTC which may be precursor to ATC.
Incidence in ATC
—
TP53
Clinical implications
Considered late stage in the carcinogenesis process. More commonly found in ATC.
Incidence in ATC
Found in 50–70% of ATC.
TERT
Clinical Implications
Considered late stage in the carcinogenesis process. More commonly found in ATC.
Incidence in ATC
Seen in 65–75% of ATC.
P13K/AKT
Clinical Implications
Considered late stage in the carcinogenesis process.
Incidence in ATC
30–40% of ATC.
EIF1AX
Clinical Implications
Often present with RAS mutations.
Incidence in ATC
—
None of the above mutations, although found in ATCs, is diagnostic for ATC.